Researchers identify a route to create surface states with a maximal energy difference between electrons with different spins.
The surfaces of materials can host unique electronic properties, where the electrons behave very differently from the interior. In particular, the intrinsically broken symmetries of the surface compared with the bulk of the sample allow for a separation of energy states according to their spin. This effect could be utilised in novel electronic devices which operate with both the charge and spin of electrons- but it is first necessary to understand how to maximise the magnitude of the effect.
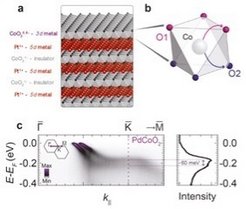
In a recent publication in Nature, researchers from the University of St Andrews and the Max Planck Institute for Chemical Physics of Solids in Dresden, in collaboration with beamline staff from Diamond Light Source (UK) and Elettra (Italy), described a new route to maximise the spin-splitting of surface states. By considering the hierarchies of energy scales in the system, they identified that a maximal spin-splitting situation could be realised by first having a very large energy scale associated with the inversion symmetry breaking at the surface. They then measured the electronic structure of PtCoO2, PdCoO2 and PdRhO2, all belonging to the delafossite family of layered oxide materials and found a significant spin-splitting, proving that this scenario can indeed be realised. Their result gives a useful principle for the design of new materials with interesting and potentially useful surfaces and interface states.
The work was supported by the European Research Council (via the QUESTDO project), the UK Engineering and Physical Sciences Research Council, the Royal Society, the Max-Planck Society, and the International Max-Planck Partnership for Measurement and Observation at the Quantum Limit.
VS ; HR / CPfS